REGKIRONA TO BE DISTRIBUTED
입력 2021.02.17 (15:36)
수정 2021.02.17 (16:51)
읽어주기 기능은 크롬기반의
브라우저에서만 사용하실 수 있습니다.
[Anchor Lead]
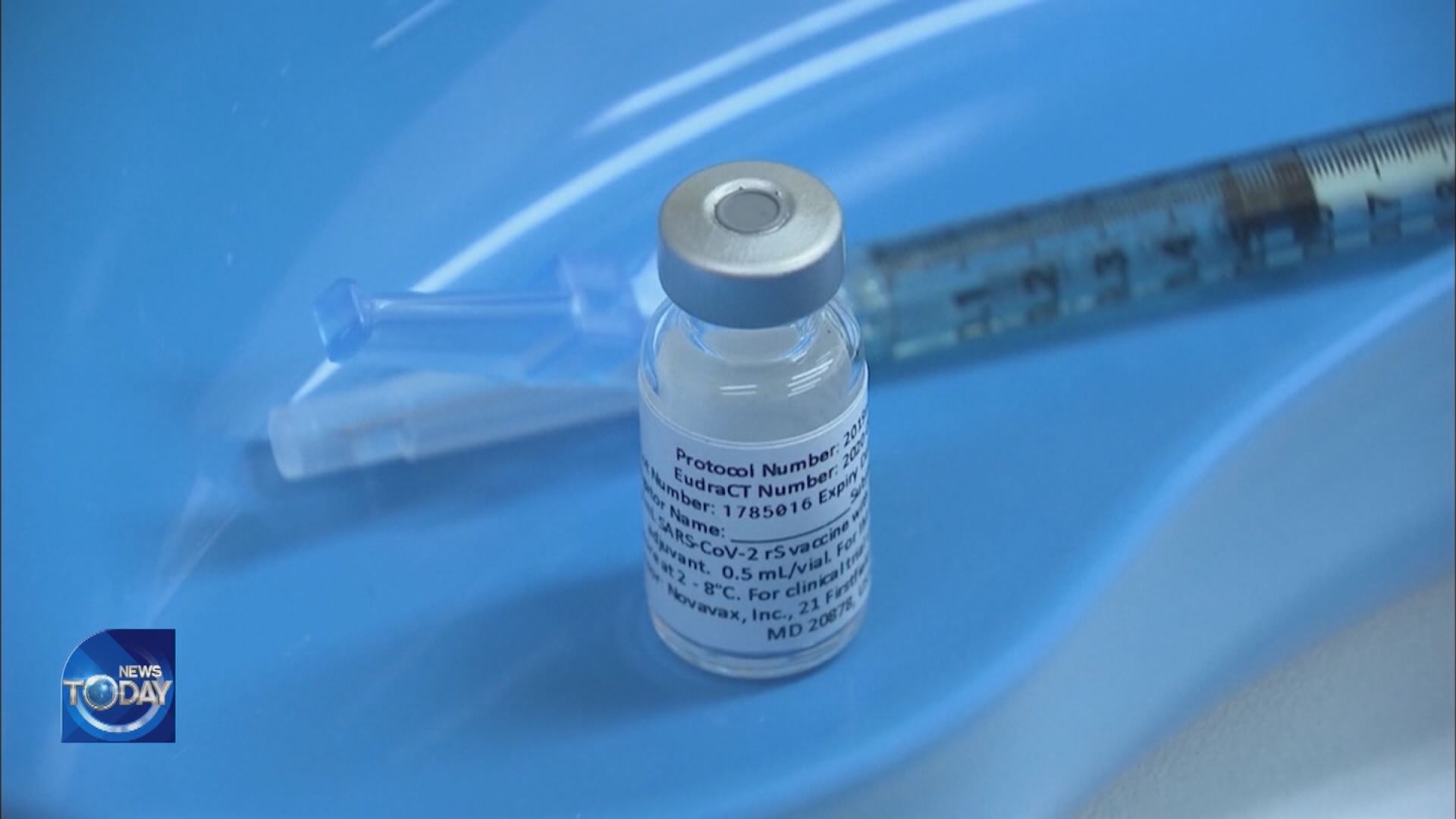
Regkirona, Korea’s first antibody therapeutic drug for COVID-19 approved by the government, will start to be distributed to medical institutions today. The Korea Disease Control and Prevention Agency announced that for a limited time the agency had purchased 960 milligrams of COVID-19 antibodies which will be made available to medical facilities today. The drug will be administered to COVID-19 patients not on ventilators who are 60 or older or with underlying conditions and whose symptoms started showing less than seven days ago.
Regkirona, Korea’s first antibody therapeutic drug for COVID-19 approved by the government, will start to be distributed to medical institutions today. The Korea Disease Control and Prevention Agency announced that for a limited time the agency had purchased 960 milligrams of COVID-19 antibodies which will be made available to medical facilities today. The drug will be administered to COVID-19 patients not on ventilators who are 60 or older or with underlying conditions and whose symptoms started showing less than seven days ago.
■ 제보하기
▷ 카카오톡 : 'KBS제보' 검색, 채널 추가
▷ 전화 : 02-781-1234, 4444
▷ 이메일 : kbs1234@kbs.co.kr
▷ 유튜브, 네이버, 카카오에서도 KBS뉴스를 구독해주세요!
- REGKIRONA TO BE DISTRIBUTED
-
- 입력 2021-02-17 15:36:53
- 수정2021-02-17 16:51:46

[Anchor Lead]
Regkirona, Korea’s first antibody therapeutic drug for COVID-19 approved by the government, will start to be distributed to medical institutions today. The Korea Disease Control and Prevention Agency announced that for a limited time the agency had purchased 960 milligrams of COVID-19 antibodies which will be made available to medical facilities today. The drug will be administered to COVID-19 patients not on ventilators who are 60 or older or with underlying conditions and whose symptoms started showing less than seven days ago.
Regkirona, Korea’s first antibody therapeutic drug for COVID-19 approved by the government, will start to be distributed to medical institutions today. The Korea Disease Control and Prevention Agency announced that for a limited time the agency had purchased 960 milligrams of COVID-19 antibodies which will be made available to medical facilities today. The drug will be administered to COVID-19 patients not on ventilators who are 60 or older or with underlying conditions and whose symptoms started showing less than seven days ago.
이 기사가 좋으셨다면
-
좋아요
0
-
응원해요
0
-
후속 원해요
0














이 기사에 대한 의견을 남겨주세요.